Isotopes_of_neon
Isotopes of neon
Nuclides with atomic number of 10 but with different mass numbers
Neon (10Ne) possesses three stable isotopes: 20
Ne
, 21
Ne
, and 22
Ne
. In addition, 17 radioactive isotopes have been discovered, ranging from 15
Ne
to 34
Ne
, all short-lived. The longest-lived is 24
Ne
with a half-life of 3.38(2) min. All others are under a minute, most under a second. The least stable is 15
Ne
with a half-life of 770(300) ys (7.7(3.0)×10−22 s). See isotopes of carbon for notes about the measurement. Light radioactive neon isotopes usually decay to fluorine or oxygen, while heavier ones decay to sodium.
Quick Facts Main isotopes, Decay ...
| ||||||||||||||||||||||||||
| Standard atomic weight Ar°(Ne) | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Close
More information Nuclide, Z ...
| Nuclide [n 1] |
Z | N | Isotopic mass (Da)[4] [n 2][n 3] |
Half-life[1] [resonance width] |
Decay mode[1] [n 4] |
Daughter isotope [n 5] |
Spin and parity[1] [n 6] |
Natural abundance (mole fraction) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Excitation energy | Normal proportion[1] | Range of variation | |||||||||||||||||
| 15 Ne [5] |
10 | 5 | 15.043170(70) | 770(300) ys [590(230) keV] |
2p | 13 O |
(3/2−) | ||||||||||||
| 16 Ne |
10 | 6 | 16.025751(22) | > 5.7 zs [< 80 keV] |
2p | 14 O |
0+ | ||||||||||||
| 17 Ne [n 7] |
10 | 7 | 17.0177140(4) | 109.2(6) ms | β+p (94.4(2.9)%) | 16 O |
1/2− | ||||||||||||
| β+α (3.51(1)%) | 13 N | ||||||||||||||||||
| β+ (2.1(2.9)%) | 17 F | ||||||||||||||||||
| β+pα (0.014(4)%) | 12 C | ||||||||||||||||||
| 18 Ne |
10 | 8 | 18.0057087(4) | 1664.20(47) ms | β+ | 18 F |
0+ | ||||||||||||
| 19 Ne |
10 | 9 | 19.00188091(17) | 17.2569(19) s | β+ | 19 F |
1/2+ | ||||||||||||
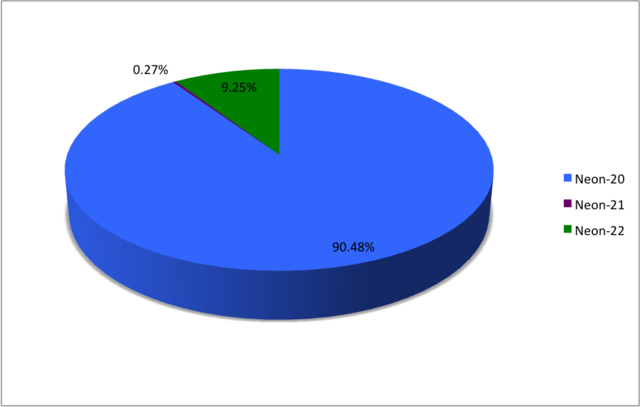
| 20 Ne |
10 | 10 | 19.9924401753(16) | Stable | 0+ | 0.9048(3) | [0.8847, 0.9051][6] | ||||||||||||
| 21 Ne |
10 | 11 | 20.99384669(4) | Stable | 3/2+ | 0.0027(1) | [0.0027, 0.0171][6] | ||||||||||||
| 22 Ne |
10 | 12 | 21.991385114(19) | Stable | 0+ | 0.0925(3) | [0.0920, 0.0996][6] | ||||||||||||
| 23 Ne |
10 | 13 | 22.99446691(11) | 37.15(3) s | β− | 23 Na |
5/2+ | ||||||||||||
| 24 Ne |
10 | 14 | 23.9936106(6) | 3.38(2) min | β− | 24m Na |
0+ | ||||||||||||
| 25 Ne |
10 | 15 | 24.997810(30) | 602(8) ms | β− | 25 Na |
1/2+ | ||||||||||||
| 26 Ne |
10 | 16 | 26.000516(20) | 197(2) ms | β− (99.87(3)%) | 26 Na |
0+ | ||||||||||||
| β−n (0.13(3)%) | 25 Na | ||||||||||||||||||
| 27 Ne |
10 | 17 | 27.007570(100) | 30.9(1.1) ms | β− (98.0(5)%) | 27 Na |
(3/2+) | ||||||||||||
| β−n (2.0(5)%) | 26 Na | ||||||||||||||||||
| β−2n ?[n 8] | 25 Na ? | ||||||||||||||||||
| 28 Ne |
10 | 18 | 28.012130(140) | 18.8(2) ms | β− (84.3(1.1)%) | 28 Na |
0+ | ||||||||||||
| β−n (12(1)%) | 27 Na | ||||||||||||||||||
| β−2n (3.7(5)%) | 26 Na | ||||||||||||||||||
| 29 Ne |
10 | 19 | 29.019750(160) | 14.7(4) ms | β− (68.0(5.1)%) | 29 Na |
(3/2−) | ||||||||||||
| β−n (28(5)%) | 28 Na | ||||||||||||||||||
| β−2n (4(1)%) | 27 Na | ||||||||||||||||||
| 30 Ne |
10 | 20 | 30.024990(270) | 7.22(18) ms | β− (78.1(4.6)%) | 30 Na |
0+ | ||||||||||||
| β−n (13(4)%) | 29 Na | ||||||||||||||||||
| β−2n (8.9(2.3)%) | 28 Na | ||||||||||||||||||
| 31 Ne |
10 | 21 | 31.033470(290) | 3.4(8) ms | β− | 31 Na |
(3/2−) | ||||||||||||
| β−n ?[n 8] | 30 Na ? | ||||||||||||||||||
| β−2n ?[n 8] | 29 Na ? | ||||||||||||||||||
| 32 Ne |
10 | 22 | 32.039720(540)# | 3.5(9) ms | β− | 32 Na |
0+ | ||||||||||||
| β−n ?[n 8] | 31 Na ? | ||||||||||||||||||
| β−2n ?[n 8] | 30 Na ? | ||||||||||||||||||
| 33 Ne ?[n 9] |
10 | 23 | 33.049520(640)# | < 260 ns | n ?[n 8] | 32 Ne |
7/2−# | ||||||||||||
| 34 Ne |
10 | 24 | 34.056730(550)# | 2 ms# [> 1.5 μs] | β− ?[n 8] | 34 Na |
0+ | ||||||||||||
| β−2n ?[n 8] | 32 Ne ? | ||||||||||||||||||
| β−n ?[n 8] | 33 Ne ? | ||||||||||||||||||
| This table header & footer: | |||||||||||||||||||
Close
- mNe – Excited nuclear isomer.
- ( ) – Uncertainty (1σ) is given in concise form in parentheses after the corresponding last digits.
- # – Atomic mass marked #: value and uncertainty derived not from purely experimental data, but at least partly from trends from the Mass Surface (TMS).
- Modes of decay:
n: Neutron emission p: Proton emission - Bold symbol as daughter – Daughter product is stable.
- ( ) spin value – Indicates spin with weak assignment arguments.
- Decay mode shown is energetically allowed, but has not been experimentally observed to occur in this nuclide.
- This isotope has not yet been observed; given data is inferred or estimated from periodic trends.
- The isotopic composition refers to that in air.
- Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties" (PDF). Chinese Physics C. 45 (3): 030001. doi:10.1088/1674-1137/abddae.
- Prohaska, Thomas; Irrgeher, Johanna; Benefield, Jacqueline; Böhlke, John K.; Chesson, Lesley A.; Coplen, Tyler B.; Ding, Tiping; Dunn, Philip J. H.; Gröning, Manfred; Holden, Norman E.; Meijer, Harro A. J. (2022-05-04). "Standard atomic weights of the elements 2021 (IUPAC Technical Report)". Pure and Applied Chemistry. doi:10.1515/pac-2019-0603. ISSN 1365-3075.
- Wang, Meng; Huang, W.J.; Kondev, F.G.; Audi, G.; Naimi, S. (2021). "The AME 2020 atomic mass evaluation (II). Tables, graphs and references*". Chinese Physics C. 45 (3): 030003. doi:10.1088/1674-1137/abddaf.
- Wamers, F.; Marganiec, J.; Aksouh, F.; Aksyutina, Yu.; Álvarez-Pol, H.; Aumann, T.; Beceiro-Novo, S.; Boretzky, K.; Borge, M. J. G.; Chartier, M.; Chatillon, A.; Chulkov, L. V.; Cortina-Gil, D.; Emling, H.; Ershova, O.; Fraile, L. M.; Fynbo, H. O. U.; Galaviz, D.; Geissel, H.; Heil, M.; Hoffmann, D. H. H.; Johansson, H. T.; Jonson, B.; Karagiannis, C.; Kiselev, O. A.; Kratz, J. V.; Kulessa, R.; Kurz, N.; Langer, C.; Lantz, M.; Le Bleis, T.; Lemmon, R.; Litvinov, Yu. A.; Mahata, K.; Müntz, C.; Nilsson, T.; Nociforo, C.; Nyman, G.; Ott, W.; Panin, V.; Paschalis, S.; Perea, A.; Plag, R.; Reifarth, R.; Richter, A.; Rodriguez-Tajes, C.; Rossi, D.; Riisager, K.; Savran, D.; Schrieder, G.; Simon, H.; Stroth, J.; Sümmerer, K.; Tengblad, O.; Weick, H.; Wimmer, C.; Zhukov, M. V. (4 April 2014). "First Observation of the Unbound Nucleus 15Ne" (PDF). Physical Review Letters. 112 (13): 132502. doi:10.1103/PhysRevLett.112.132502. PMID 24745409 – via APS.
- Meija, Juris; Coplen, Tyler B.; Berglund, Michael; Brand, Willi A.; Bièvre, Paul De; Gröning, Manfred; Holden, Norman E.; Irrgeher, Johanna; Loss, Robert D.; Walczyk, Thomas; Prohaska, Thomas (2016-03-01). "Isotopic compositions of the elements 2013 (IUPAC Technical Report)". Pure and Applied Chemistry. 88 (3): 293–306. doi:10.1515/pac-2015-0503. hdl:11858/00-001M-0000-0029-C408-7. ISSN 1365-3075. S2CID 104472050.